NPSD Dashboards
The Agency for Healthcare Research and Quality (AHRQ) presents the dashboards of patient safety data received for analysis and publication in the Network of Patient Safety Databases (NPSD). The NPSD dashboards were initially published in June 2019 and were based on more than 1.1 million records reported by healthcare providers to approximately 15 percent of AHRQ-listed Patient Safety Organizations (PSOs) through March 31, 2018. Since then, the dashboards have been updated annually with new data submitted to the PSO Privacy Protection Center. Providers and PSOs that are willing to contribute data play an essential role in growing the NPSD into an ever-more-robust resource for patient safety and quality improvement.
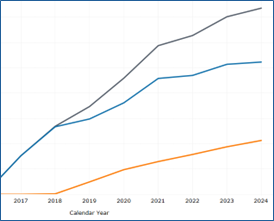
Two sets of interactive hospital safety incident dashboards are presented: the Data Submission Summary Dashboard, which provides a high-level overview of the frequency of patient safety events reported; and the Patient Safety Event Dashboards, which include a generic section that provides an overview of the numbers and categories of various patient safety event reports and event-specific dashboards that describe in greater depth specific safety events such as medication events and falls. Through the interactive functions, users can choose to view the dashboards reflecting the more than 3.6 million cumulative records submitted through 2024 in Common Formats for Event Reporting – Hospitals version 1.2 and 2.0.
Hospital Safety Concerns Dashboards
Data Submission Dashboard
Examines all reports on patient safety concerns submitted to the PSOPPC. The dashboard charts detail reports submitted by Common Formats version by year, completeness of reports submitted by version, percentage of reports by version, percentage of reports by report type, and percentage of events by event type and version.
Generic Dashboard
Is based on general information gathered from reports of patient safety concerns associated with at least one of ten specific event types. The dashboard charts detail event type, report type by event type, extent of harm by event type, event type by extent of harm, and extent of harm.
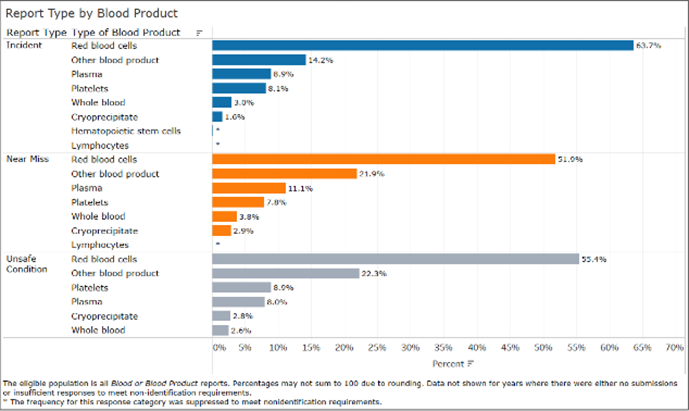
Blood and Blood Product Dashboard
Details the type of blood product involved, the report type, report type by blood product, reported contributing factor to the event, the incorrect action, and the extent of patient harm by age category.
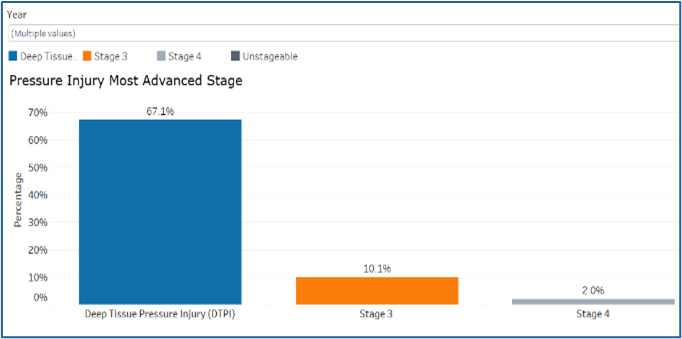
Pressure Injury Dashboard
Details the documentation of pressure Injury most advanced stage, residual patient harm level, extent of harm by most advanced stage, contributing factor, extent of harm by contributing factor, documented skin inspection on admission, documented increased risk for pressure injury following a risk assessment; preventive intervention implemented, extent of harm by preventive intervention and extent of harm by age category.
Device or Medical/Surgical Supply
Details the type of device; type of device by residual harm to the patient; device defect, failure, or user error; device defect, failure, or user error by residual harm to the patient; type of health information technology (HIT) device; and type of HIT device by residual harm to the patient.
Falls Dashboard
Details the extent of harm due to falls, the presence of fall assistance, presence of fall assistance by patient harm, type of fall injury, and fall prevention.
Medication or Other Substance Dashboard
Includes information organized by medication or other substance events at a glance, including description of safety concerns, stage of care concern originated, and type of medication/substance involved for incidents, near misses, and unsafe conditions. The dashboard then further examines incidents and near misses along with their contributing factors and preventability.
Perinatal Dashboard
Details whether originated during either the birthing process or an intrauterine procedure.
Learn more about how the individual dashboards are set up.
Additional Notes
- As only data submitted in the Common Formats for Event Reporting – Hospitals (CFER-H) are included in the NPSD dashboards, the dashboards are characterized as reflecting data from the hospital setting. While it is believed that the CFER-H are primarily used as intended to capture patient safety events in hospital settings, providers may have used the CFER-H to report data from other settings.
- Each individual dashboard presents the responses to particular data elements. The primary dashboard is a figure depicting the relative response rates, which is paired with a data table presenting the specific data points. Details on the frequency and percentage of responses can be seen by hovering the mouse over points of interest. Captions provide details about the eligible population and other information necessary to interpret the figure.
- Drop-down menus are provided where there is sufficient data to allow the user to filter and select the data along specific parameters.
- Technical notes follow each dashboard to provide additional details such as the text of the specific Common Formats question on which the dashboard is based, any constraints on the eligible population of reports for the data element (such as whether the question is limited to certain report types), and the data element response rate and population size.